Research
Each subcellular compartment of the eukaryotic cell can be characterized by typical redox conditions. Redox environment is tightly associated with the redox reactions of the intermediary metabolism, therefore they represent a base of cellular regulation, sensing, and signaling. The redox conditions within a compartment are mainly defined by the quantitatively most significant local redox reactions, but they can be influenced also by the transmembrane transport of redox-active compounds and the transmembrane electron fluxes. Redox-active compounds are usually small electron carriers functioning as cofactors/substrates of oxidoreductases. The redox state of these couples can be regarded as an indicator of the redox environment of the cell or an organelle. Moreover, major redox pairs can constitute redox buffers that, on one hand soothe sudden changes in redox conditions, on the other hand define the oxidative or reductive character of the compartment. The lumen of the endoplasmic reticulum (ER) constitutes a separate intracellular compartment with a special proteome and metabolome. The redox conditions of the organelle are characteristically different from those of the other subcellular compartments. A special feature of the luminal environment is a split: redox systems in reduced and oxidized states are present simultaneously. The concerted action of membrane transporters and luminal oxidoreductases maintains the oxidized state of the thiol/disulfide and the reduced state of the pyridine nucleotide redox systems, which are prerequisites for the normal redox reactions localized in the organelle.
Similarly to other subcellular organelles, the major redox buffers in the endoplasmic reticulum lumen are the thiol/disulfide and the reduced/oxidized pyridine nucleotide couples. The most striking feature of the ER lumen is the oxidized state of the thiol/disulfide system, which results in a redox potential of approximately -180 mV, much higher than that of the cytosol (-230 mV). Consequently, the lumen of the ER has been regarded as an oxidizing environment, in agreement with the fact that the oxidative protein folding - a major pathway of the compartment - requires oxidizing power. Recent observations indicate that pyridine nucleotides are overwhelmingly present in a reduced form in the ER lumen. Luminal NADPH is required for the prereceptorial activation of glucocorticoids, for some reactions of biotransformation and presumably for local antioxidant defense. Genetic or pharmacological manipulation of the luminal NADPH generation profoundly affects local glucocorticoid effects and initiates apoptotic or autophagic mechanisms. The powerful thiol-oxidizing machinery of oxidative protein folding (a major pathway in the lumen) continuously challenges the local antioxidant defense in professional secretory cells. Alterations of the luminal redox conditions, either in oxidizing or reducing direction, affect protein processing, are sensed by the accumulation of misfolded/unfolded proteins, and may induce ER stress and unfolded protein response. The activated signaling pathways attempt to restore the balance between protein loading and processing and induce programmed cell death if these attempts fail. Recent findings strongly support the involvement of redox-based endoplasmic reticulum stress in a plethora of human diseases, either as causative agents or as complications. Luminal redox systems can also act as sensors for electron donors and acceptors ( i.e. nutrients and oxygen); this way, the ER has a central role in the fine-tuning of environmental and internal stimuli. Redox imbalance affecting any redox system finally leads to ER stress and initiates ER-dependent signaling pathways to restore the physiological conditions. Exhaustion of the protective mechanisms results in various ER-dependent forms of programmed cell death. The pathological consequences of ER redox imbalance constitutes important factor in the pathomechanism of widespread human diseases such as neurodegenerative and cardiovascular diseases, metabolic diseases, and cancer.
Grants
OTKA-K 100293 - Bánhegyi Gábor - GLUT 10 deficiency and the arterial tortuosity syndrome
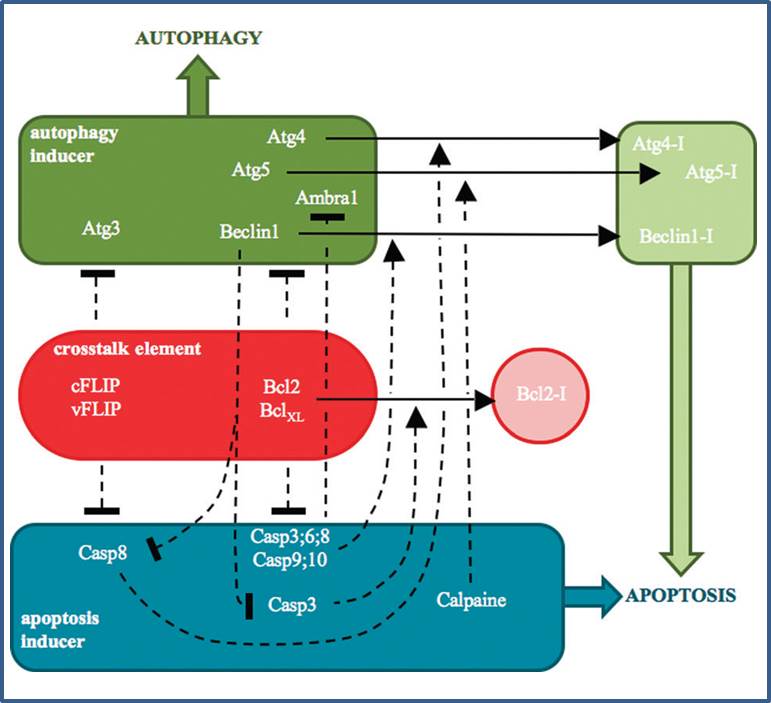
OTKA-PD 104110 - Kapuy Orsolya - A systems biological view of choosing between autophagy and apoptosis after endoplasmic reticulum stress
Lab Head

Gábor Bánhegyi, MD, PhD
email: banhegyi.gabor@med.semmelweis-univ.hu
phone: (36 1) 459 15 00/60101
Researchers
Graduate students
Undergraduate students
Mariann Holczer
Éva Keczán
Laura Kiss
Gergely János Kosztyi
Margita Márton
Publications
2013
The Inter-Relationship of Ascorbate Transport, Metabolism and Mitochondrial, Plastidic Respiration. Szarka A, Bánhegyi G, Asard H. Antioxid Redox Signal. 2013 Feb 13.
Minireview: endoplasmic reticulum stress: control in protein, lipid, and signal homeostasis. Mandl J, Mészáros T, Bánhegyi G, Csala M. Mol Endocrinol. 2013 Mar;27(3):384-93. doi: 10.1210/me.2012-1317. Epub 2013 Jan 24.
A cellular stress-directed bistable switch controls the crosstalk between autophagy and apoptosis. Kapuy O, Vinod PK, Mandl J, Bánhegyi G. Mol Biosyst. 2013 Feb 2;9(2):296-306. doi: 10.1039/c2mb25261a. Epub 2012 Dec 7.
2012
Multiple roles of glucose-6-phosphatases in pathophysiology: State of the art and future trends. Marcolongo P, Fulceri R, Gamberucci A, Czegle I, Banhegyi G, Benedetti A. Biochim Biophys Acta. 2012 Dec 21;1830(3):2608-2618. doi: 10.1016/j.bbagen.2012.12.013.
The glucose-6-phosphate transport is not mediated by a glucose-6-phosphate/phosphate exchange in liver microsomes. Marcolongo P, Fulceri R, Giunti R, Margittai E, Banhegyi G, Benedetti A. FEBS Lett. 2012 Sep 21;586(19):3354-9. doi: 10.1016/j.febslet.2012.07.018. Epub 2012 Jul 20.
Production of H2O2 in the endoplasmic reticulum promotes in vivo disulfide bond formation. Margittai É, Löw P, Stiller I, Greco A, Garcia-Manteiga JM, Pengo N, Benedetti A, Sitia R, Bánhegyi G. Antioxid Redox Signal. 2012 May 15;16(10):1088-99. doi: 10.1089/ars.2011.4221.
The endoplasmic reticulum as the extracellular space inside the cell: role in protein folding and glycosylation. Csala M, Kereszturi É, Mandl J, Bánhegyi G. Antioxid Redox Signal. 2012 May 15;16(10):1100-8. doi: 10.1089/ars.2011.4227. Epub 2012 Feb 23. Review.
G6PT-H6PDH-11ßHSD1 triad in the liver and its implication in the pathomechanism of the metabolic syndrome. Czegle I, Csala M, Mandl J, Benedetti A, Karádi I, Bánhegyi G. World J Hepatol. 2012 Apr 27;4(4):129-38. doi: 10.4254/wjh.v4.i4.129.
Guidelines for the use and interpretation of assays for monitoring autophagy. Klionsky DJ, et al. (Bánhegyi G). Autophagy. 2012 Apr;8(4):445-544.
2011
Inhibition of glycoprotein synthesis in the endoplasmic reticulum as a novel anticancer mechanism of (-)-epigallocatechin-3-gallate. Konta L, Száraz P, Magyar JÉ, Révész K, Bánhegyi G, Mandl J, Csala M. Biofactors. 2011 Nov-Dec;37(6):468-76. doi: 10.1002/biof.189. Epub 2011 Dec 8.Expression of hexose-6-phosphate dehydrogenase in rat tissues. Marcolongo P, Senesi S, Giunti R, Csala M, Fulceri R, Bánhegyi G, Benedetti A. J Steroid Biochem Mol Biol. 2011 Sep;126(3-5):57-64. doi: 10.1016/j.jsbmb.2011.05.006. Epub 2011 May 19.
Enhanced activity of galactono-1,4-lactone dehydrogenase and ascorbate-glutathione cycle in mitochondria from complex III deficient Arabidopsis. Zsigmond L, Tomasskovics B, Deák V, Rigó G, Szabados L, Bánhegyi G, Szarka A. Plant Physiol Biochem. 2011 Aug;49(8):809-15. doi: 10.1016/j.plaphy.2011.04.013. Epub 2011 May 7.
Overexpression limits of fission yeast cell-cycle regulators in vivo and in silico. Moriya H, Chino A, Kapuy O, Csikász-Nagy A, Novák B. Mol Syst Biol. 2011 Dec 6;7:556. doi: 10.1038/msb.2011.91.
Switches and latches: a biochemical tug-of-war between the kinases and phosphatases that control mitosis. Domingo-Sananes MR, Kapuy O, Hunt T, Novak B. Philos Trans R Soc Lond B Biol Sci. 2011 Dec 27;366(1584):3584-94. doi: 10.1098/rstb.2011.0087. Review.
Protein phosphatase 2A controls the order and dynamics of cell-cycle transitions. Krasinska L, Domingo-Sananes MR, Kapuy O, Parisis N, Harker B, Moorhead G, Rossignol M, Novák B, Fisher D. Mol Cell. 2011 Nov 4;44(3):437-50. doi: 10.1016/j.molcel.2011.10.007.
System-level feedbacks make the anaphase switch irreversible. He E, Kapuy O, Oliveira RA, Uhlmann F, Tyson JJ, Novák B. Proc Natl Acad Sci U S A. 2011 Jun 14;108(24):10016-21. doi: 10.1073/pnas.1102106108. Epub 2011 May 26.
2010
Decreased prereceptorial glucocorticoid activating capacity in starvation due to an oxidative shift of pyridine nucleotides in the endoplasmic reticulum. Kereszturi É, Kálmán FS, Kardon T, Csala M, Bánhegyi G. FEBS Lett. 2010 Nov 19;584(22):4703-8. doi: 10.1016/j.febslet.2010.10.053. Epub 2010 Oct 28.
Contribution of fructose-6-phosphate to glucocorticoid activation in the endoplasmic reticulum: possible implication in the metabolic syndrome. Senesi S, Legeza B, Balázs Z, Csala M, Marcolongo P, Kereszturi E, Szelényi P, Egger C, Fulceri R, Mandl J, Giunti R, Odermatt A, Bánhegyi G, Benedetti A. Endocrinology. 2010 Oct;151(10):4830-9. doi: 10.1210/en.2010-0614. Epub 2010 Sep 8.
Oxidative folding in the endoplasmic reticulum: towards a multiple oxidant hypothesis? Margittai E, Bánhegyi G. FEBS Lett. 2010 Jul 16;584(14):2995-8. doi: 10.1016/j.febslet.2010.05.055. Epub 2010 May 31. Review.
Redox control of endoplasmic reticulum function. Csala M, Margittai E, Bánhegyi G. Antioxid Redox Signal. 2010 Jul 1;13(1):77-108. doi: 10.1089/ars.2009.2529. Review.
BGP-15 inhibits caspase-independent programmed cell death in acetaminophen-induced liver injury. Nagy G, Szarka A, Lotz G, Dóczi J, Wunderlich L, Kiss A, Jemnitz K, Veres Z, Bánhegyi G, Schaff Z, Sümegi B, Mandl J. Toxicol Appl Pharmacol. 2010 Feb 15;243(1):96-103. doi: 10.1016/j.taap.2009.11.017. Epub 2009 Nov 26.
Hexose-6-phosphate dehydrogenase in the endoplasmic reticulum. Senesi S, Csala M, Marcolongo P, Fulceri R, Mandl J, Banhegyi G, Benedetti A. Biol Chem. 2010 Jan;391(1):1-8. doi: 10.1515/BC.2009.146. Review.
Altered redox state of luminal pyridine nucleotides facilitates the sensitivity towards oxidative injury and leads to endoplasmic reticulum stress dependent autophagy in HepG2 cells. Száraz P, Bánhegyi G, Benedetti A. Int J Biochem Cell Biol. 2010 Jan;42(1):157-66. doi: 10.1016/j.biocel.2009.10.004. Epub 2009 Oct 9.
Life and death of a BiP substrate. Otero JH, Lizák B, Hendershot LM. Semin Cell Dev Biol. 2010 Jul;21(5):472-8. doi: 10.1016/j.semcdb.2009.12.008. Epub 2009 Dec 21. Review.
Systems-level feedback in cell-cycle control. Novák B, Vinod PK, Freire P, Kapuy O. Biochem Soc Trans. 2010 Oct;38(5):1242-6. doi: 10.1042/BST0381242. Review. Erratum in: Biochem Soc Trans. 2010 Dec;38(6):1723.
Regulated protein kinases and phosphatases in cell cycle decisions. Novak B, Kapuy O, Domingo-Sananes MR, Tyson JJ. Curr Opin Cell Biol. 2010 Dec;22(6):801-8. doi: 10.1016/j.ceb.2010.07.001. Epub 2010 Aug 2. Review.